In quantum mechanics, spin is an intrinsic form of angular momentum carried by elementary particles such as electrons.
The water we drink every day is made of hydrogen and oxygen atoms. In a simple model of a hydrogen atom, an electron is bound to a proton. Because atoms are far too small to picture directly, we will use an “everyday scale” to express atomic dimensions in more familiar terms.
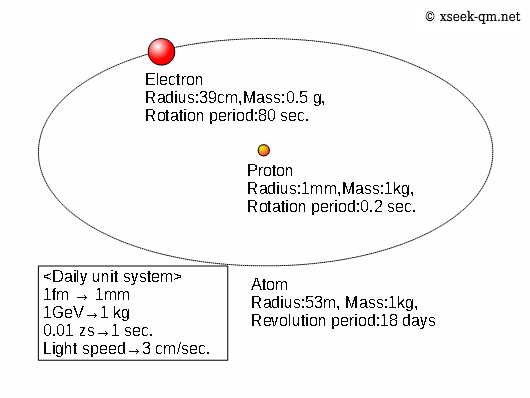
In this figure, the electron’s radius is shown as 390 femtometers, which corresponds to 39 centimeters on the everyday scale. In reality, experiments show that the electron’s radius is smaller than 1 femtometer, or less than 1 millimeter on the same scale.
Protons and electrons both have intrinsic spin. In quantum mechanics, this means that they possess a fixed amount of angular momentum.
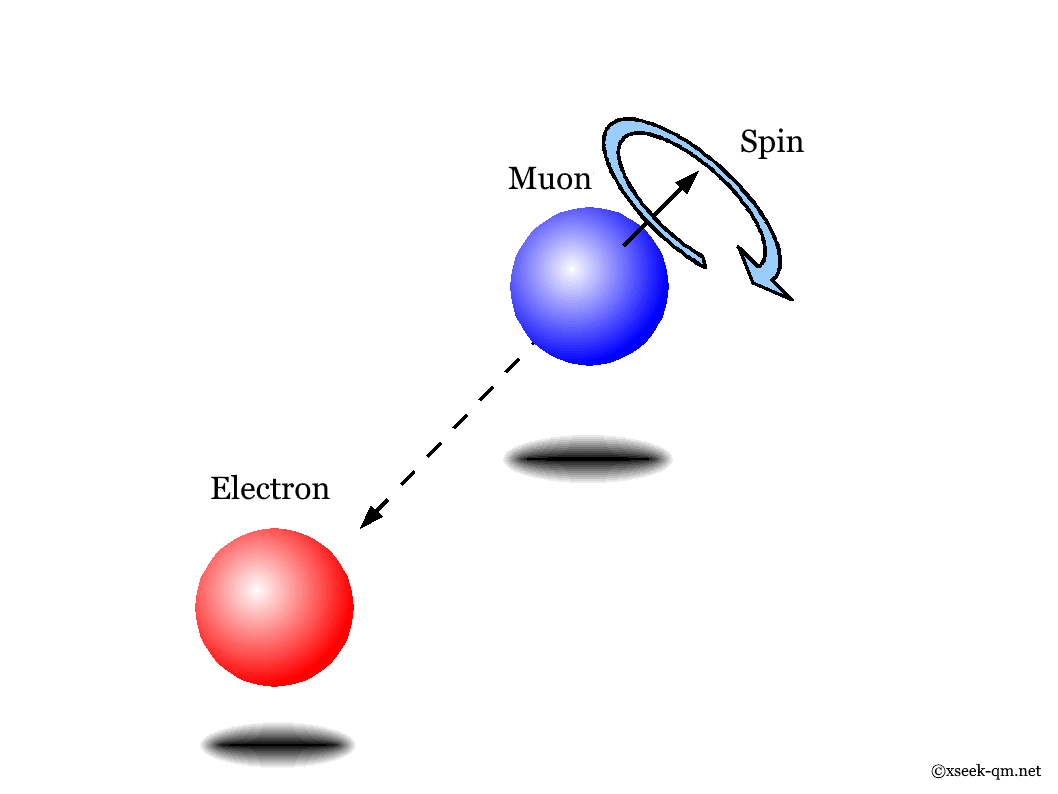
To understand spin more concretely, consider the muon, a particle similar to the electron. Its mass is 106 MeV, which corresponds to 106 grams on the everyday scale, and its lifetime is about two microseconds, or roughly 700 million years on that scale. A muon decays into an electron and two neutrinos.

Without a magnetic field, the spin axis can point in any direction. Its direction can be inferred by observing the decay electron, which tends to be emitted opposite to the muon’s spin.
In a magnetic field, however, the spin can take only two orientations: parallel or anti-parallel to the field. The spin state is a superposition of these two possibilities, and the direction in which the decay electron is emitted reflects that state. In this sense, the magnetic field breaks the system’s rotational symmetry.
Many people say that spin is not a literal rotation. There are two main reasons for this view.
Experiments show that the electron’s radius is less than 1 femtometer, or less than 1 millimeter on the everyday scale. If we tried to explain its spin angular momentum as ordinary classical rotation, the electron’s surface would have to move at about 390 times the speed of light. Even if we used the much larger radius of 390 femtometers, or 39 centimeters on the everyday scale, the required surface speed would still be equal to the speed of light.
A 360° rotation changes the sign of a spin-½ wave function. Only after a 720° rotation does the wave function return to its original phase. For this reason, the spin wave function is said to be double-valued.
Because of these difficulties, many physicists do not regard spin as ordinary rotation. To support a rotational interpretation of spin, I wrote the following paper.
I hope this paper offers a helpful way to understand spin as a kind of rotational phenomenon.
© 2002-2014 xseek-qm.net
広告


