This page presents visualizations of the electron wave functions of the hydrogen atom.
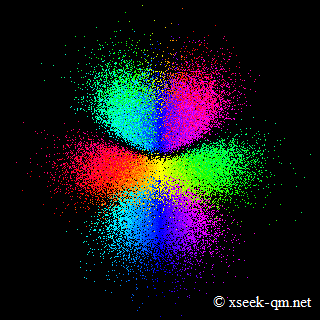
In Bohr's model of the atom, electrons are pictured as moving around the nucleus in fixed orbits. In modern quantum-mechanical descriptions of atomic orbitals, such as the visualization below, electrons are not depicted as particles traveling along definite paths.
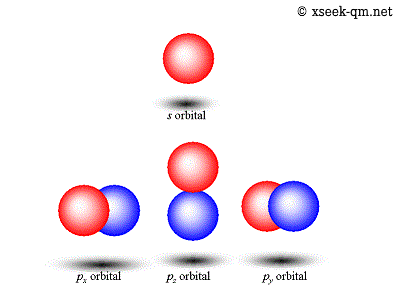
In the second row, the image suggests a relationship between the red sphere and the orbital's angular momentum, but it does not make the axis of rotation clear.
By contrast, the "Wave Functions Illustrated by Phase" page of the Mathematical Science Art Museum visualizes wave functions in the following way:
This approach makes it possible to animate the apparent motion of electrons around the nucleus.
I therefore experimented with creating these visualizations in JavaScript.
A GIF animation of the rotating electron wave function for the 4f orbital of the hydrogen atom is available here:
The animation can also be viewed on YouTube.
The video was produced using the following steps:
Changing only the phase makes the wave function appear to rotate.
You can also try a JavaScript-based animation of the electron wave function in a hydrogen atom here:
The default state is (n, l, m) = (4, 3, 1). Here, n is the principal quantum number, l is the azimuthal quantum number, and m is the magnetic quantum number.
Click the "State" button to choose from states such as (1, 0, 0), (2, 1, 1), (3, 2, 1), and (3, 2, 2). In the (1, 0, 0) state, no rotation occurs; only the global phase changes.
The default number of points is 51,200. If your computer cannot run the animation smoothly, click the "Number" button to reduce the number of points.
© 2012-2013 xseek-qm.net
広告


