This site is intended as an entry point into the world of quantum theory. The articles are written so that even readers with little background in physics can follow them. I hope they spark your interest in the strange and fascinating world of quantum theory.
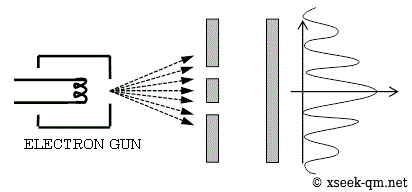
Let us begin with one of the most famous experiments in quantum mechanics: the double-slit experiment.

In the figure above, electrons are fired toward a barrier with two slits. Each electron either hits the barrier or passes through one of the slits. Electrons that pass through then strike a screen behind the barrier. What kind of pattern appears when many electrons pass through the slits? At first, as shown in the next figure, only a few individual dots appear on the screen.
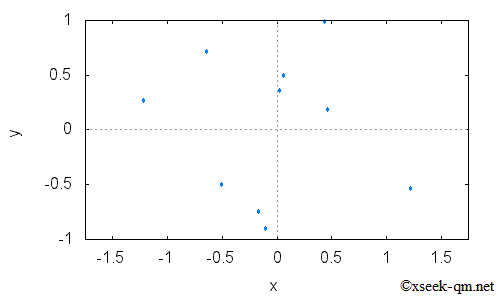
As more electrons are fired, however, the distribution gradually changes as follows.
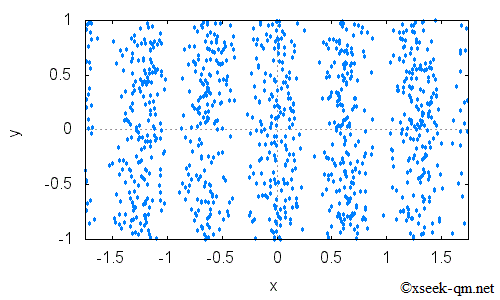
A striped interference pattern appears on the screen. When the electron distribution is plotted as a graph, it looks like this:
These stripes appear because electrons have wave-like properties. Waves passing through the two slits overlap, sometimes reinforcing each other and sometimes canceling each other out. This phenomenon is called interference, and it produces the striped pattern. This is the essential mystery of the double-slit experiment. What makes it so puzzling is that even when electrons pass through the apparatus one at a time, they still eventually form an interference pattern.
The electron's wave is described by a function such as the following:
This function is called the wave function and is pronounced "psi of x." The variable x represents the electron's position. In quantum mechanics, the wave function can take complex values and describes the state of the electron.
In the double-slit experiment, the electron is associated with two possible paths: one through the right slit and one through the left slit. We denote the wave function for the path through the right slit by ΨR(x) and the wave function for the path through the left slit by ΨL(x). The total wave function is obtained by adding these two wave functions:
This addition of wave functions is called superposition, and it leads to interference. The probability P(x) of detecting the electron at position x is given by the square of the magnitude of the wave function:
Why is the detection probability given by the square of the wave function's magnitude?
In 1965, physicist Richard Feynman wrote the following in The Feynman Lectures on Physics, Volume III:
One might still like to ask: "How does it work? What is the machinery behind the law?"
No one has found any machinery behind the law. No one can "explain" any more than we have just "explained." No one will give you any deeper representation of the situation. We have no ideas about a more basic mechanism from which these results can be deduced.
In other words, no deeper mechanism is currently known. The textbook Introduction to Gauge Theory also quotes Einstein as writing in 1951:
All the fifty years of conscious brooding have brought me no closer to the answer to the question, "What are light quanta?" Of course today every rascal thinks he knows the answer, but he is deluding himself.
Through this site, I hope we can explore the wonders of quantum theory together.
© 2002, 2014 xseek-qm.net
広告


